Many protozoa live in the human body. Many of them are pathogenic. Our story is about a dozen of them, the most-most. The review is based on historical and recent publications.
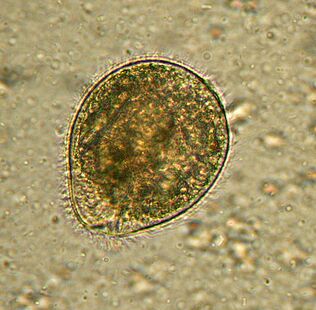
Largest. BalantidiumBalantidium coli
The largest protozoan is a human parasite and is the only ciliary in this company. Its dimensions vary from 30 to 150 microns in length and from 25 to 120 microns in width. For comparison: the length of Plasmodium falciparum in the highest stage is about 15 microns and several times smaller than the balantidia of intestinal cells, among which live ciliates. An elephant in a Chinese shop.
Distributedwherever there are pigs - its main carriers. It usually lives in the submucosa of the colon, although in humans it also occurs in the pulmonary epithelium. It feeds on bacteriaB. coli, food particles, fragments of host epithelium. In animals, the infection is asymptomatic. People can develop severe diarrhea with bloody, mucous secretions (balantidiasis), sometimes ulcers form on the walls of the colon. It rarely dies from balantidiasis, but it causes chronic exhaustion.
People are infected with dirty water or food that contains cysts. The infection rate in humans does not exceed 1%, while pigs can be infected worldwide.
Treatedwith antibiotics, no drug resistance reports have been reported for this ciliary body.
Discoveredby the Swedish scientist Malstem in 1857. Today, balantidiasis is associated with tropical and subtropical areas, poverty and poor hygiene.
First. Oral amoebaEntamoeba gingivalis
The first parasitic amoeba found in humans. The description of the amoeba was published in 1849 in the oldest scientific journal. An amoeba was found in dental plaque, hence the name from the Latin gingiva - gum.
Livesin the mouth of almost all people with sore teeth or sore gums, inhabits gum pockets and plaque. It feeds on epithelial cells, leukocytes, microbes, and in the case of erythrocytes. It is rare in people with a healthy oral cavity.
This small protozoan, 10–35 µm in size, does not enter the environment and does not form cysts, it is transmitted to another host by kissing, dirty dishes or contaminated food. E. gingivalisis considered to be exclusively a human parasite, but is sometimes found in captive cats, dogs, horses, and monkeys.
In the early twentieth century,E. gingivaliswas described as the cause of periodontal disease because it is always present in inflamed tooth cells. However, its pathogenicity has not been proven.
The drugsthat affect this amoeba are unknown.
Most pervasive. Dysentery amoebaEntamoeba histolytica
This intestinal parasite with blood penetrates the tissues of the liver, lungs, kidneys, brain, heart, spleen, genitals. It eats what it will get: food particles, bacteria, red blood cells, leukocytes and epithelial cells.
Distributedeverywhere, especially in the tropics. Usually people become infected by swallowing a cyst.
In temperate countries the amoeba tends to remain in the intestinal lumen and the infection is asymptomatic. In tropical and subtropical regions, the pathological process often begins:E. histolyticaattacks the walls. The reasons for the transition to the pathogenic form are not yet clear, but several molecular mechanisms of what is happening have already been described. Thus, it is clear that amoebae secrete lysis substances, break through mucus, and kill cells. Apparently, an amoeba can destroy a host cell in two ways: by initiating apoptosis in it or simply by chewing pieces. The first method has long been considered the only one. Otherwise, the mechanism of cell suicide at record speed - in a few minutes - has not been identified. Another method was described recently, the authors called it trogocytosis from the Greek "troika" - to bite. Significantly, amoebae that bite cells leave their prey as soon as they die. Others can phagocytose dead cells completely. It is assumed that the cells that bite and devour differ in the pattern of gene expression.
Now the ability of the amoeba to penetrate the bloodstream, liver and other organs is associated with trogocytosis.
Amoeba is a deadly disease, about 100 thousand people die each year from infectionE. histolytica.
The dysenteric amoeba has a non-pathogenic twin,E. dispar, so microscopy is not sufficient to diagnose the disease.
To be curedmust be destroyed as mobileE. histolyticaand cysts.
DescribedE. histolyticaand its pathogenic nature was established in 1875 in a patient with diarrhea. The Latin name for the amoeba was given in 1903 by the German zoologist Fritz Schaudin.Histolyticameans destructive tissue. In 1906, the scientist died of an amoebic bowel abscess.
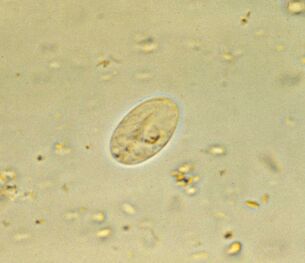
Most common. Intestinal lambliaGiardia lamblia (G. intestinalis)
Giardia, the most common intestinal parasite, is ubiquitous. 3-7% of people in developed countries and 20-30% in developing countries are infected. That's about 300 million people.
Parasites livein the duodenum and bile ducts of the host, where they either swim, working with whips, or attach to the epithelium using an adhesive disc located on the underside of the cell. In 1 cm2, the epithelium adheres to one million lamblia. They damage the villi, which interferes with the absorption of nutrients, causing inflammation of the mucous membranes and diarrhea. If the disease affects the bile ducts, it is accompanied by jaundice.
Giardiasis is a disease of dirty hands, water and food. The life cycle of protozoa is simple: in the intestine there is an active form, and at the exit with the fecal masses of a stable cyst. To become infected, it is enough to swallow a dozen cysts, which will turn into the active form in the intestines again.
The main secretof the ubiquity of lamblia in the variability of surface proteins. The human body fights lamblia with antibodies and, in principle, is capable of developing immunity. But people who live in the same area and drink the same water re-infect the offspring of their own parasites. Why? Because during the transition from the active phase to the cyst and vice versa, lamblia changes the proteins to which antibodies are produced - surface proteins specific to the variant. There are about 190 variants of these proteins in the genome, but only one is always present on the surface of a single parasite, the translation of the remainder interrupting the mechanism of RNA interference. And change happens about once every ten generations.
It is treatedwith an antiprotozoal agent with antibacterial action. The disease passes in a week, but if the bile ducts are infected, relapses are possible for many years. Cysts fight with iodizing water.
DiscoveredGiardia lambliaIn 1859 by the Czech scientist Willem Lambl. Since then, it has changed several names in the simplest way, and the current one was given in honor of the discoverer and French parasitologist Alfred Giar, who did not describe lamblia.
And the first sketch of Giardia was made by Anthony van Leeuwenhoek, who found it in his upset chair. It was 1681.
By the way, Giardia is also very evolutionarily ancient, coming almost directly from the ancestor of all eukaryotes.
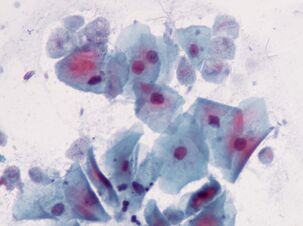
Most intimate. Trichomonas vaginalisTrichomonas vaginalis.
The simplest, sexually transmitted. It lives in the vagina, and in men - in the urethra, epididymis and prostate, it is transmitted sexually or with wet cloths. Babies can become infected by passing through the birth canal.T. vaginalishas 4 whips at the anterior end and a relatively short wavy membrane; if necessary, it releases pseudopods. The maximum size of a trichomonas is 32 by 12 microns.
Trichomonas ismore prevalentthan the causative agents of chlamydia, gonorrhea and syphilis combined. It affects about 10% of women, and maybe more, and 1% of men. The latter figure is unreliable because it is more difficult to detect parasites in men.
T. vaginalisfeeds on microorganisms, including lactic acid bacteria of the vaginal microflora, which maintain an acidic environment, thus creating an optimal pH for itself above 4, 9.
Trichomonas destroys mucosal cells, causing inflammation. About 15% of infected women complain of symptoms.
Treatedwith antibacterial drug. As a preventive measure, regular watering with diluted vinegar is recommended.
Describedin 1836 by the French bacteriologist Alfred Donne. The scientist did not understand that there was a pathogenic parasite in front of him, but he determined the size, appearance and type of movement of the simplest.
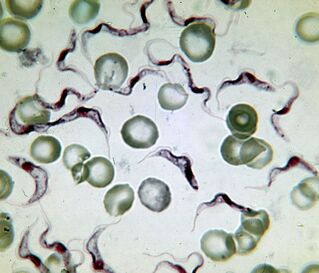
Most deadly. Cause of sleeping sicknessTrypanosoma brucei
The cause of African sleep sickness is the deadliest protozoan. A person infected with it dies without treatment. Trypanosoma is an elongated whip 15-40 μm long. There are two known subspecies that cannot be distinguished externally. Disease causedT. brucei gambiense, lasts 2-4 years.T. brucei rhodesienseis a more virulent, transient pathogen from which they die after a few months or weeks.
Distributedin Africa, between the 15th parallel of the Southern and Northern Hemispheres, in the natural range of the carrier - blood-sucking insects of the genusGlossina(cece fly). Of the 31 species of flies, 11 are dangerous to humans. Sleeping sickness affects the population of 37 countries south of the Sahara at 9 million km2. Up to 20, 000 people get sick every year. There are now about 500, 000 patients, 60 million living in danger.
From the intestinal intestineT. bruceienters the human bloodstream, thence into the cerebrospinal fluid and affects the nervous system. The disease begins with fever and inflammation of the lymph glands, followed by lethargy, drowsiness, muscle paralysis, exhaustion, and irreversible coma.
The lethality of the parasite is related to its ability to cross the blood-brain barrier. Molecular mechanisms are not fully understood, but it is known that when it enters the brain, the parasite secretes cysteine proteases and also uses some host proteins. On the other hand, in the central nervous system, trypanosomes are confined to immune factors.
The first description of sleep disorders in the upper reaches of the Niger was left by the Arab scholar Ibn Khaldun (1332-1406). At the beginning of the 19th century, Europeans were already aware of the initial sign of the disease - swelling of the lymph nodes in the back of the head (a symptom of Winterbottom), and slave traders paid special attention to it.
DiscoveredT. bruceiScottish microbiologist David Bruce, after whom it was named, and in 1903 he first established a link between trypanosomes, bed bugs and sleeping sickness.
Treatmentdepends on the stage of the disease, and medications cause serious side effects. The parasite has high antigenic variability, so it is impossible to create a vaccine.
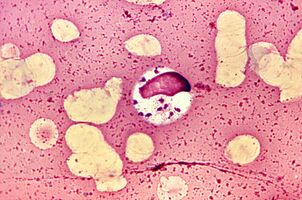
The most extravagant. LeishmaniaLeishmania donovani
Leishmans deserve the title of the most extravagant parasites, because they live and multiply in macrophages - cells designed to destroy parasites.L. donovaniis the most dangerous of them. It causes visceral leishmaniasis, colloquially dumdum fever or kala azar, from which almost all patients die without treatment. But survivors acquire long-term immunity.
There are three subspecies of parasites.L. donovani infantum(Mediterranean and Central Asia) primarily affects children and is often a reservoir for dogs.L. donovani donovani(India and Bangladesh) is dangerous for adults and the elderly, there are no natural reservoirs. AmericanL. donovani chagasi(Central and South America) can live in the blood of dogs.
L. donovani- whip not more than 6 microns long. People become infected after being bitten by mosquitoes of the genusPhlebotomus, sometimes through sexual contact, babies - passing through the birth canal. Once in the blood,L. donovanipenetrate macrophages, which transmit the parasite through the internal organs. Propagating in macrophages, the parasite destroys them. The molecular mechanism of survival in macrophages is quite complex.
Symptoms of the disease- fever, enlarged liver and spleen, anemia and leukopenia, which contribute to secondary bacterial infection. Every year, 500 thousand people get visceral leishmaniasis and about 40 thousand die.
Treatmentdifficult - intravenous transfusion of antimony and blood.
Taxonomic affiliationL. donovaniwas established in 1903 by the famous malaria researcher and Nobel laureate Ronald Ross. He owes his generic name to William Leishman, and especially to Charles Donovan, who in 1903 independently discovered protozoan cells in the spleen of patients who died of Azara feces, one in London, the other in Madras.
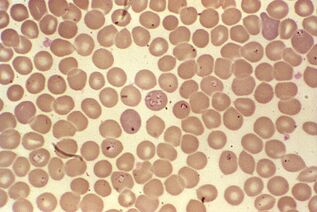
The hardest life cycle. Babesia spp.
Babesia, in addition to multistage asexual reproduction in mammalian erythrocytes and sex mites in the intestines of the genusIxodes, complicated their development by transovarian transmission. From the intestines of female mites, protozoan sporozoites penetrate the ovaries and infect embryos. When the mite larvae hatch, the babesia pass into their salivary glands and enter the vertebrate blood with the first bite.
DistributedBabesia in America, Europe and Asia. Their natural reservoirs are rodents, dogs and cattle. A person is infected with several species: B. microti, B. divergens, B. duncaniandB. venatorum.
The symptoms of babesiosis are similar to malaria - recurrent fever, hemolytic anemia, enlarged spleen and liver. Most people recover spontaneously; for patients with a weakened immune system, babesiosis is fatal.
Treatment methodsare still being developed, while antibiotics and, in severe cases, blood transfusions are prescribed.
Babesia was described by the Romanian microbiologist Victor Babes (1888), who discovered it in sick cows and sheep. He decided he was dealing with a pathogenic bacterium he namedHaematococcus bovis. Babesia was long considered an animal pathogen, until it was discovered in 1957 in a Yugoslav shepherd who died of B. divergens infection.
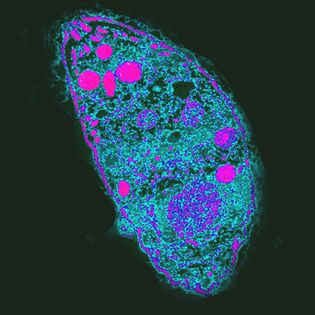
Most influential. Cause of ToxoplasmosisToxoplasma gondii
T. gondiiis the most powerful parasite because it controls the behavior of middle hosts.
Distributedeverywhere, unevenly distributed. For example, in France, 84% of the population is infected, in the United Kingdom - 22%.
The life cycle of toxoplasma consists of two phases: asexual occurs in the body of any warm-blooded, sexual reproduction is possible only in the epithelial cells of the feline intestine. ThatT. gondiicould complete development, the cat must eat an infected rodent. Increasing the likelihood of this event,T. gondiiblocks rodents' natural fear of the smell of cat urine and makes it an attractive targeting group of neurons in the amygdala. How it works is not known. One of the putative mechanisms of action is the local immune response to infection. It changes the level of cytokines, which in turn raises the level of neuromodulators such as dopamine. Toxoplasma also affects human behavior, which is manifested even at the population level. Thus, in countries with high levels of toxoplasmosis, neuroticism and a desire to avoid precarious new situations are more common. It is possible that infection withT. gondiicould lead to cultural change.
Infectionin humans is often asymptomatic, but with weakened immunity it destroys cells of the liver, lungs, brain, retina, causing acute or chronic toxoplasmosis. The course of infection depends on the virulence of the strain, the state of the host's immune system and its age - older people are less susceptibleT. gondii.
Treattoxoplasmosis with antiprotozoal drugs.
Describedin 1908 in desert rodents. This honor belongs to the staff of the Pasteur Institute in Tunisia, Charles Nicolas and Luis Mans.
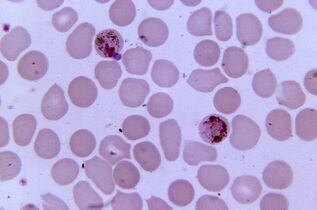
The most pathogenic. Plasmodium malariaPlasmodium spp.
Plasmodium malaria is the most pathogenic parasite in humans. The number of people suffering from malaria can reach 300-500 million, and the mortality rate during epidemics - 2 million. This disease still takes three times more lives than armed conflicts.
Five species of Plasmodium cause malaria in humans:Plasmodium vivax, P. falciparum, P. malariae, P. ovaleandP. knowlesi, which also affects macaques.
Distributedin the vector range - mosquitoesAnopheles, which require a temperature of 16–34 ° C and a relative humidity of more than 60%.
A comparison of the genome of the most virulent of the plasmodia,P. falciparum, and the plasmodia of the gorilla suggests that humans were infected by his ancestor from these monkeys. The appearance of this form of plasmodium is connected with the appearance of agriculture in Africa, which led to an increase in population density and the development of irrigation systems.
Sexual reproduction of plasmodia occurs in the intestines of mosquitoes, and in the human body it is an intracellular parasite that lives and multiplies in hepatocytes and erythrocytes until the cells rupture. 1 ml of the patient's blood contains 1 - 50 thousand parasites.
The disease manifests itself as inflammation, recurrent fever and anemia, in case of pregnancy it is dangerous for the mother and fetus. Erythrocytes infected with P. falciparum clog the capillaries, and in severe cases, ischemia of internal organs and tissues develops.
Treatmentrequires a combination of several drugs and depends on the specific pathogen. Plasmodia become resistant to drugs.






































